This Week on Life Sciences Digital
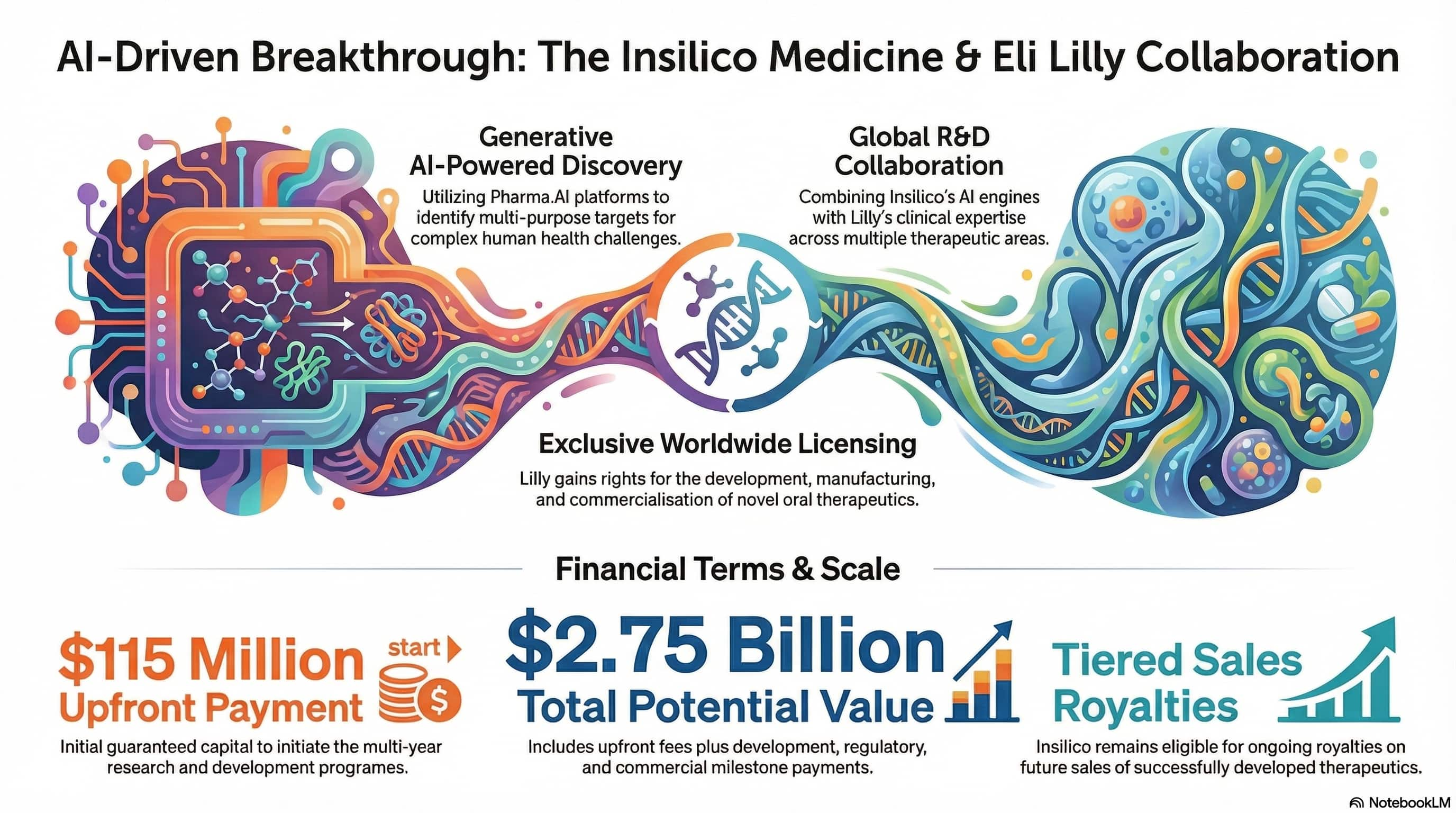
Insilico Medicine announced a global drug discovery collaboration with Eli Lilly, granting Lilly an exclusive worldwide license for the development, manufacturing, and commercialization of a portfolio of novel oral therapeutics in preclinical development across multiple therapeutic areas. Under the terms of the agreement, Insilico is eligible to receive a $115 million upfront payment, with total deal value reaching approximately $2.75 billion in development, regulatory, and commercial milestones, plus tiered royalties on future sales.
This is not a research partnership or a pilot agreement. It is $115 million in cash before a single clinical result, from a top-5 pharma company, on an exclusive license — structured like an acquisition without the acquisition. The deal signals something the industry has been debating for three years: whether AI-native drug discovery platforms can generate assets that major pharma will pay for at scale. Lilly just answered that question.
Beyond the licensed portfolio, Insilico and Lilly will collaborate on multiple R&D programs targeting indications selected by Lilly, combining Insilico's Pharma.AI platforms with Lilly's development capabilities and deep disease-area expertise. Insilico is the only AI drug discovery company with its own asset in clinical trials — ISM001-055, currently in Phase 2 for idiopathic pulmonary fibrosis. That track record is almost certainly what unlocked the upfront.
The context makes this landing harder: Lilly committed $1 billion to an NVIDIA AI lab in January. It is now committing $2.75 billion in potential value to an external AI discovery platform in March. Lilly is not choosing between building and buying AI capability. It is doing both, at speed. For every AI drug discovery company watching the M&A landscape — this deal sets the new reference price.
Visual summary generated with AI (NotebookLM).
Also This Week:
Caris Life Sciences launched the Caris AI Insights™ signature for pancreatic ductal adenocarcinoma — built on its CodeAI™ platform, integrated into the standard Caris Molecular Tumor Board Report, and deployable without collecting additional tissue. The tool guides therapy selection between FOLFIRINOX and gemcitabine/nab-paclitaxel using whole exome and transcriptome sequencing data, and risk-stratifies patients for treatment de-escalation or more intensive regimens.
This matters for one reason that precedes all others: pancreatic cancer has a five-year survival rate below 15%, and first-line therapy selection is currently driven by clinical factors that routinely produce suboptimal outcomes. An AI tool that risk-stratifies at the molecular level before the treatment decision is made — without burdening the patient with additional procedures — changes the starting position of every clinical conversation. Caris processes millions of molecular tests annually and is already embedded in oncology workflows across major health systems.
Tempus AI signed a partnership with Daiichi Sankyo to integrate its multimodal AI models and real-world oncology database into active clinical development for an antibody drug conjugate — covering patient stratification and clinical development workflows. The distinction from previous Tempus partnerships is structural: this is not data licensing, it is embedded co-development. Tempus is operating inside the drug pipeline, not alongside it. While the stock is down 25% year-to-date, this likely reflects a sector-wide correction for AI-pure-plays rather than the underlying strength of these high-value clinical partnerships.
Belén Garijo published concrete enterprise AI adoption figures this week: Merck's internal conversational AI platform, myGPT — the first deployed by a DAX-listed company — now has 34,000 monthly active users. The company's unified analytics platform sees 4,000 weekly users. The No. 2 ranking in the Global Pharma AI Readiness Index is less interesting than what it represents: AI adoption inside pharma is now measurable, comparable, and tracked as a competitive metric.
Signals & Market Moves
In a Databricks session, Merck and ZS detailed the deployment of agentic AI using AI/BI Genie and Agent Bricks: domain-specific data agents operating on governed, semantically enriched business data to automate complex analytical workflows across brand performance and marketing operations. Result: over 50% reduction in time to generate commercial insights, with measurable improvements in decision consistency.
The Signal: This is agentic AI running in pharma commercial operations — not in R&D, not in a pilot, but in the business units that run product launches. Merck appears twice in this issue for a reason.
Governed AI Becomes a Legal Requirement, Not Just a Best Practice 🔗
An Observer analysis this week made the governance case from the security angle: traditional compliance frameworks — ISO 27001, SOC 2 — were not designed for machine learning operations. As AI embeds into drug discovery and clinical development, new risk categories emerge: model training data exposure, adversarial exploitation, MLOps-specific attack surfaces. The EU AI Act and the FDA's evolving guidance on AI-enabled medical devices are tightening requirements on transparency and security across AI systems in regulated workflows.The Signal: Two weeks ago, a federal court ruled that documents prepared using public generative AI platforms lose attorney-client privilege — because third-party AI tools carry no expectation of confidentiality. Last week, we covered shadow AI governance in R&D labs. This week, the security infrastructure layer gets the same scrutiny. The pattern is consistent: governed, auditable, sovereign AI is no longer a differentiator. It is table stakes, and regulators are catching up to where the market already is.
Tool Spotlight from our Life Sciences Digital database
AI-powered scientific data catalog and knowledge platform.
Got a tool for life sciences you’d like more people to know?
Events & Calls
Impact Challenge: AI for Science
The $30M global open call from Google.org is still accepting applications until April 17, 2026. Selected organizations receive between $500K and $3M, plus six months of pro bono technical support from Google experts and Google Cloud credits. Focus areas include AI for Health & Life Sciences. For research-stage organizations and nonprofits, this is one of the most accessible funding mechanisms in the market right now.
🤝 Got a tool for life sciences you'd like more people to know about?
Submit your tool here.
Want to be featured in a future issue or explore sponsorship?
We highlight the AI tools, companies, and initiatives shaping the future of life sciences. If you'd like to collaborate, reply to this email or get in touch: [email protected]




